
Secondary end points included major pathological response (≤10% residual viable tumor cells in the primary tumor and sampled lymph nodes), time to death or distant metastases, and overall survival. The other primary end point was pathological complete response (0% residual viable tumor cells in the primary tumor and sampled lymph nodes) according to blinded independent pathological review. Event-free survival was also analyzed with the use of a secondary definition, which is provided in the Methods section in the Supplementary Appendix. Event-free survival was defined as the time from randomization to any progression of disease precluding surgery, progression or recurrence of disease after surgery, progression of disease in the absence of surgery, or death from any cause data on patients with subsequent therapy were censored at the last tumor assessment that could be evaluated on or before the date of subsequent therapy. One primary end point was event-free survival according to blinded independent central review. 18 Here, we report the results of CheckMate 816, a phase 3 trial to evaluate the efficacy and safety of neoadjuvant nivolumab plus chemotherapy (three cycles) as compared with chemotherapy alone (three cycles) in patients with resectable NSCLC. 20 In addition, patients with a pathological complete response had significantly longer overall and progression-free survival than those who had an incomplete or major pathological response. 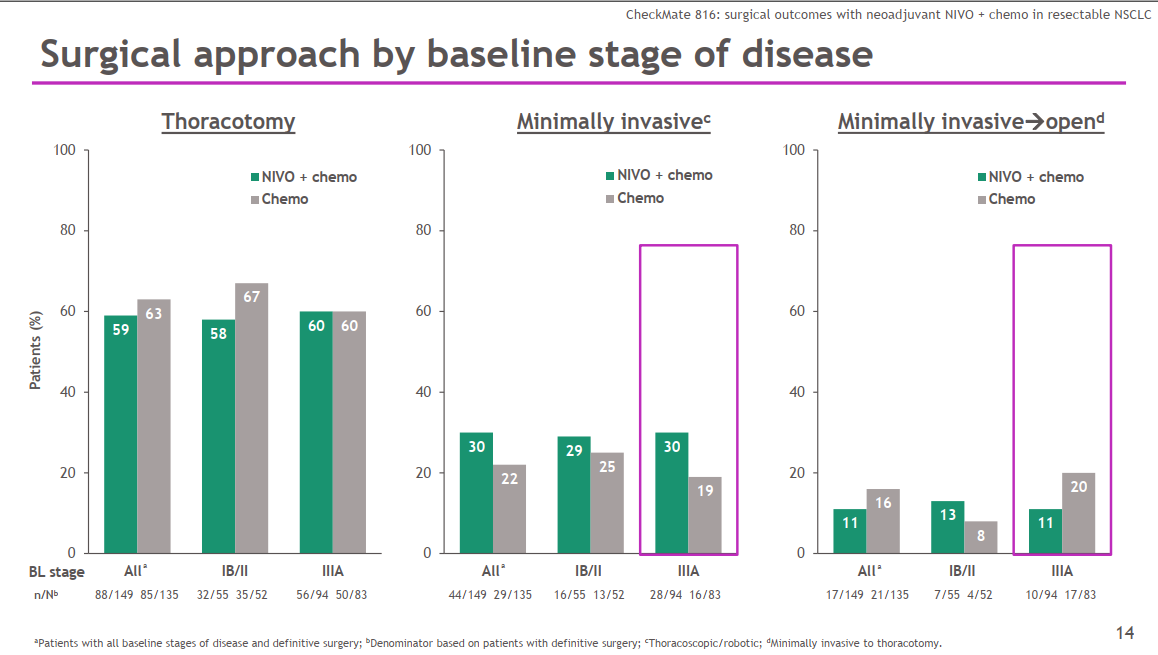
17- 19 Among patients with resectable stage IIIA NSCLC who received neoadjuvant nivolumab and chemotherapy, 3-year overall survival and progression-free survival were 81.9% and 69.6%, respectively. 15, 16 In phase 2 studies of resectable NSCLC, neoadjuvant nivolumab alone or with chemotherapy showed promise with respect to pathological complete response, survival outcomes, and safety profiles. 12- 14 In the neoadjuvant context, immunotherapy provides an early opportunity to treat micrometastatic disease and enhances the immune response when bulk tumor and tumor antigens are still present during the treatment. 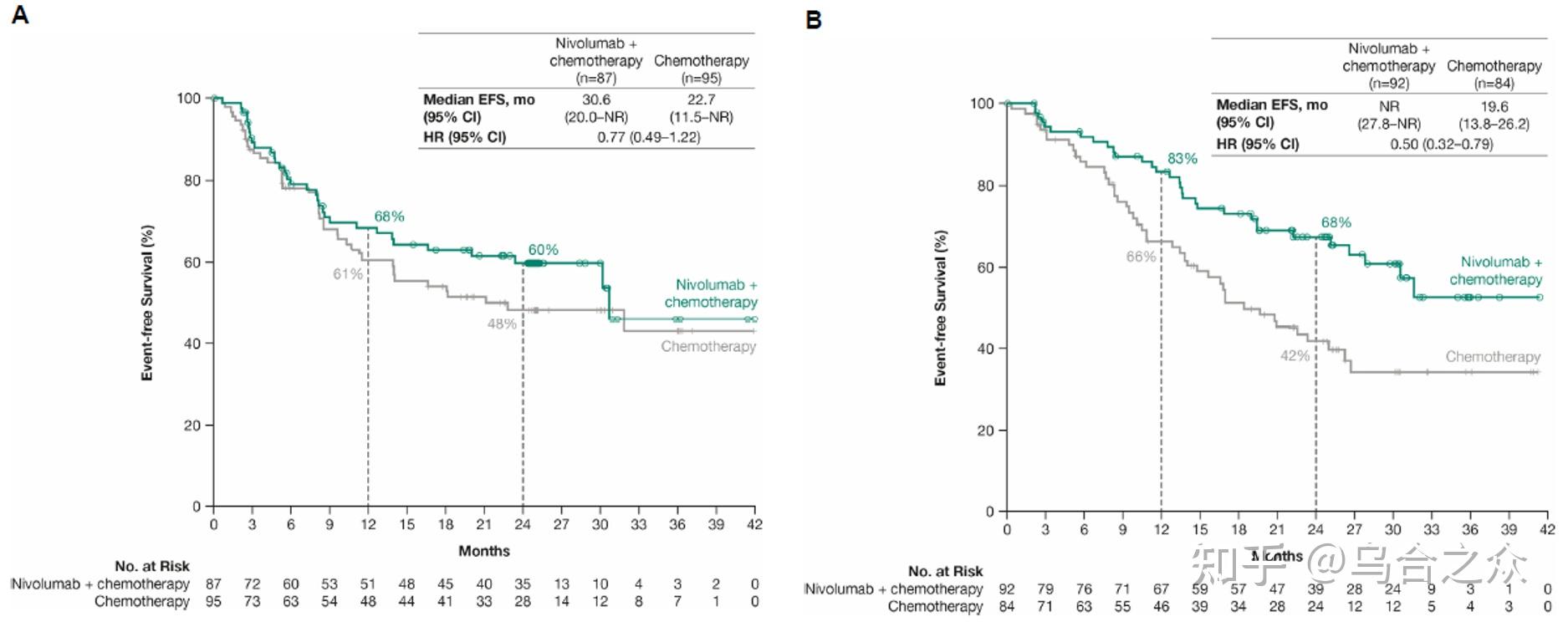
10, 11 Nivolumab-based regimens have shown a survival benefit in patients with metastatic NSCLC. Nivolumab, a fully human anti–programmed death 1 (PD-1) antibody, restores the function of existing antitumor T cells, and chemotherapy enhances antitumor immunity through direct or indirect immune-system activation. 6, 7 Although recent advances have been made with adjuvant therapies for resectable NSCLC, 8, 9 effective systemic treatments continue to be needed for nonmetastatic disease across perioperative contexts. 5 Moreover, few patients have a pathological complete response (median, 4% range, 0 to 16), a potential early predictor of survival. However, the absolute difference in 5-year recurrence-free survival and overall survival with neoadjuvant chemotherapy as compared with surgery alone is only 5 to 6 percentage points. 2, 3 Neoadjuvant chemotherapy can be used for the treatment of patients whose disease is at stages that warrant adjuvant chemotherapy (see the National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology 4 full citation provided in the Supplementary Appendix, available with the full text of this article at ). Grade 3 or 4 treatment-related adverse events occurred in 33.5% of the patients in the nivolumab-plus-chemotherapy group and in 36.9% of those in the chemotherapy-alone group.Ī pproximately 20 to 25% of patients who receive a diagnosis of non–small-cell lung cancer (NSCLC) have resectable disease 1 however, 30 to 55% of patients who undergo curative surgery have recurrence and ultimately die of their disease. Of the patients who underwent randomization, 83.2% of those in the nivolumab-plus-chemotherapy group and 75.4% of those in the chemotherapy-alone group underwent surgery. At the first prespecified interim analysis, the hazard ratio for death was 0.57 (99.67% CI, 0.30 to 1.07) and did not meet the criterion for significance. Results for event-free survival and pathological complete response across most subgroups favored nivolumab plus chemotherapy over chemotherapy alone. The median event-free survival was 31.6 months (95% confidence interval, 30.2 to not reached) with nivolumab plus chemotherapy and 20.8 months (95% CI, 14.0 to 26.7) with chemotherapy alone (hazard ratio for disease progression, disease recurrence, or death, 0.63 97.38% CI, 0.43 to 0.91 P=0.005).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
